Dry electrodes have some clear advantages but how does their signal quality compare to wet electrodes? Choosing one over the other may be a tradeoff between time, signal quality and stability.
In the previous blog post we looked at the fundamentals behind two types of EEG electrodes – Wet and Dry electrodes as well as their relative advantages . In this blog we will look at a few EEG studies that have compared the signal quality between these two electrode types.
As we mentioned in the previous blog post, dry electrodes are less time-consuming than wet electrodes. However the most important criteria is whether dry electrodes can be reliably used within the clinical environment is its data quality compared to wet electrodes. Although time-consuming, EEG recorded from wet electrodes are still considered gold standard due to the low skin contact impedance, thanks to the electrolytic gel. How does the high contact impedance of dry EEG electrodes impact the quality of EEG signals ?
Event related potentials (P3 response)
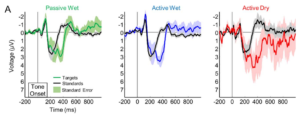
Mathewson and colleagues compared the EEG signals obtained from passive wet, active wet and active dry EEG electrodes from eight participants [1], where they completed an auditory oddball task. The figure below shows the grand average across all (artifact-free) trials obtained at the Pz electrode. We can see the expected P3 response around 250-450 ms after the presentation of the rare target stimuli in all three electrode types, however with larger standard error of mean (across trials) in the case of active dry electrode (shaded region).
Figure from [1]
Power Spectrum
They also computed the average power spectra (at electrode Pz) for each subject by averaging the FFT across the trials. Finally, the grand average power spectra across eight-participants was computed, which is shown below, with the shaded region representing the standard error of mean. Here it can be seen that the expected 1/f behavior is produced in all the electrode types. Also, the mean root mean square (RMS) values during the 200-ms baseline period (i.e. 200 ms before the presentation of the stimulus) for 10,000 permutations of 300 random standard target trials, which gives the estimate of noise on single-trial EEG epochs was computed as well. As it can be seen from the figure, the active dry EEG electrode clearly showed higher single-trial noise level compared to the active/passive wet electrodes.
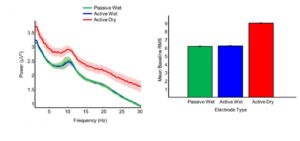
Figure from [1]
Overall the results from this study showed that active dry EEG electrodes has higher noise levels (due to higher contact impedance) compared to the wet electrodes, however they are shown to be capable of capturing the expected EEG responses, both in terms of power spectra and the ERP signature in the oddball task. Thus, despite the increased noise levels, the spatio-temporal aspects of the signal (the timing and topography) obtained with active dry EEG electrodes were very similar to those recorded using traditional wet electrodes.
EEG Sleep Spindles
A more recent study by Leach and colleagues [2] compared properties of sleep EEG recorded (from five patients) from both dry and wet (pre-gelled) electrodes. The performances were compared with respect to (1) Sleep scoring, (2) visual inspection of detected artifacts, (3) detection of important sleep characteristics such as slow waves and sleep spindles and (4) spectral characteristics and it was observed that both type of electrodes were able to reliably recorded slow waves and sleep spindles (see Figure below), with comparable signal-to-noise ratios.
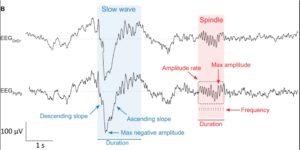
Figure from [2]
Similar conclusions about acceptable data quality from dry EEG electrodes compared to their wet counterparts have been backed by few other studies as well, showing high degree of correlation between EEG signals obtained with wet and dry electrodes [3-5], including in neonatal seizure detection [6].
Which one should you pick?
However, it has to be noted that these studies have been performed on a limited number of participants (<30) and experimental paradigms (most of them had stationary tasks). For instance, in situations where the subject is allowed to move or is prone to movements, wet electrodes are believed to be better suited as they are less prone to motion artifacts (due to low skin-electrode impedance).
Choosing the right type of electrodes depends entirely on the need. For stable long-term recordings, dry EEG electrodes are better suited as the EEG signal might be more stable over long durations, although the EEG signal itself could be noisy due to high contact impedance. Wet EEG electrodes may provide better quality EEG signals for short duration. As the gel dries up, motion artifacts and high contact impedance may deteriorate the signal further. Thus if stability is preferred over quality, dry EEG electrodes are better. On the other hand, if high quality EEG signals are required and the duration of the experiment is shorter, wet electrodes provide a good solution. But, given the rapid development in software (for artifact removal and preprocessing) as well as hardware, dry EEG electrodes are catching up in providing similar quality EEG signals.
Reference
[1] Mathewson, Kyle E., Tyler JL Harrison, and Sayeed AD Kizuk. “High and dry? Comparing active dry EEG electrodes to active and passive wet electrodes.” Psychophysiology 54.1 (2017): 74-82.
[2] Leach, Sven, et al. “A protocol for comparing dry and wet EEG electrodes during sleep.” Frontiers in neuroscience 14 (2020): 586.
[3] Hinrichs, Hermann, et al. “Comparison between a wireless dry electrode EEG system with a conventional wired wet electrode EEG system for clinical applications.” Scientific reports 10.1 (2020): 1-14.
[4]Di Flumeri, Gianluca, et al. “The dry revolution: evaluation of three different EEG dry electrode types in terms of signal spectral features, mental states classification and usability.” Sensors 19.6 (2019): 1365.
[5] Chi, Yu M., et al. “A practical mobile dry EEG system for human computer interfaces.” International Conference on Augmented Cognition. Springer, Berlin, Heidelberg, 2013.
[6] Fridman, Igor, et al. “Evaluation of dry sensors for neonatal EEG recordings.” Journal of clinical neurophysiology: official publication of the American Electroencephalographic Society 33.2 (2016): 149.


I record DC potentials. It is simply not possible to record clean potentials with dry electrodes. A simple cross-section of dry vs wet electrodes will convince you that dry electrodes are not the way to go. Further, what do you do with curly hair? In my experience, it is still required to use Ag/AgCl electrodes with a gel or paste. Any movement including EMG will inject artifact. I understand the pressure to speed up the process, but careful electrode placement using standard positioning is not hard and makes a world of difference in recording quality.
What about passive dry electrodes and in hairless surfaces?
Thanks for sharing. I enjoy reading your articles. References to ‘Dry electrodes’ should include the electrical system that backs it up. It is like comparing driving experiences of cars by focusing on just the tires. Of course dry electrodes are noisier on the front end due to the higher impedance and/or lack of skin prep. But below are some questions important to consider for a complete and accurate evaluation:
1. What if the electrode is dry to the touch yet conforms better to the skin surface thereby reducing motion artifacts? Not all dry electrodes are the same.
2. What if the electrode can be shielded from noise?
3. What if the noise can be removed dynamically and accurately?
4. What if the pre-amplification eliminates most noise pick up (common with unshielded wires with passive systems)?
5. What if the cap has been designed so as to fit well, comfortably and thereby reduce artifacts?
6. What if the electrodes are designed so as to get through challenging hair types and have good contact with the skin?
7. What if software could further help reliably remove artifacts?
These are topics that will provide the complete picture. Hopefully we can change the question asked to which ‘complete EEG solution’ is better and for what use case (electrodes+electrical_system+cap_design+software)?
I am the Founder of Zeto, Inc., and an electrical engineer by training. We sell an easy to use, FDA cleared EEG product to clinics, hospitals and research institutions. Cheers.