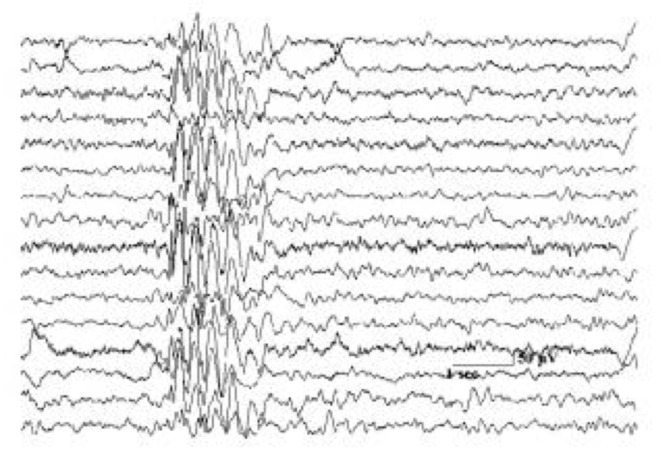
Despite a large number of papers and competitions, accurate detection of seizures remains elusive. Here are some of the key challenges.
According to the World Health Organization (WHO), approximately one-third of the patients suffering from epilepsy do not respond adequately to anti-epileptic drugs. The unpredictable nature of seizures adversely affects the quality of life in these patients. In such cases, the timely prediction of the impending seizure would be extremely beneficial for two reasons – 1) To help the patients or their caregivers to take precautionary measures against seizure-related injuries and 2) To help the research community to develop new methods to control or suppress the forthcoming seizure, for example using electrical stimulation.
However, seizure prediction has proven to be an extremely challenging task, despite the number of research papers addressing the problem growing exponentially over the past four decades.
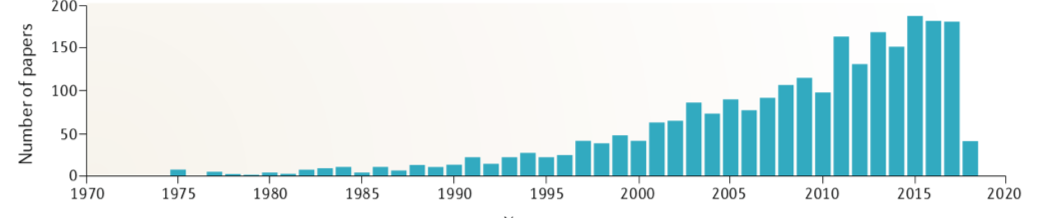
Figure 1. The number of papers published during the past three decades on seizure prediction (Data generated via PubMed using the keywords “seizure prediction”, “seizure anticipation” and “seizure forecasting”). Figure adapted from [1]
Seizure prediction pipeline
Seizure prediction algorithms typically use intracranial or scalp EEG data , to which various time (and /or frequency) analysis methods are applied to extract features , which are then fed to a decision making system (such as a classifier, for example support vector machines, artificial neural networks etc), that can separate data into two classes (for example interictal and pre-ictal). Supervised classifiers are more commonly used which makes use of labeled data, i.e. EEG datasets where pre-ictal and interictal periods are already identified by human experts. This data is then split into testing and training data. Using the training data, the classifiers learns the most optimal weights (or functions) that best separate the two classes of data. As a simple example, one can think of linear discriminant analysis (LDA) classifier, which tries to maximize the distance between the means of the two classes [2]. After identifying the optimal weights, the testing data is used to evaluate the performance of the classifier, i.e. to see how well the classifier generalizes to datasets on which it was not trained. A typical schematic of seizure prediction system is shown below
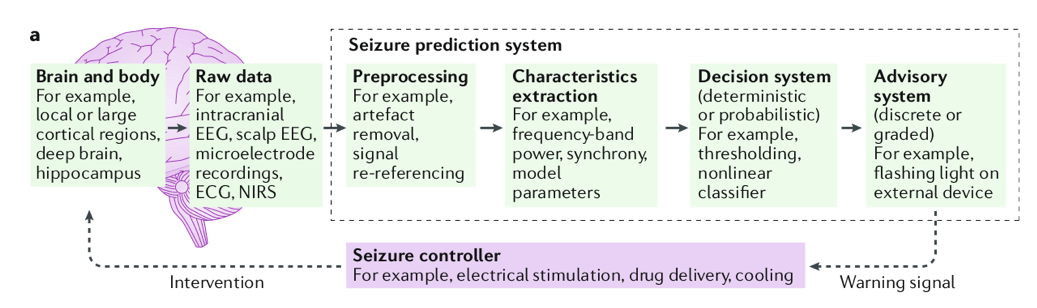 Figure 2. Typical seizure prediction system. Figure adapted from [1]
Figure 2. Typical seizure prediction system. Figure adapted from [1]
Apart from this machine learning based approach outlined above, developments in deep learning have found their way into seizure prediction algorithms. The advantage of deep learning approach is that it can handle raw data and can perform automatic feature extraction, compared to machine learning based approach which would require some data pre-processing (which in turn can affect data in a bad way) and feature extraction using time and or frequency based approaches.
Seizure Detection Competitions and Algorithms
Several seizure predicting competitions have also taken place over the past decade on Kaggle and elsewhere. These enable comparison of algorithms, which is beneficial for the field. However, this also poses a challenge in terms of the correct way of evaluating these algorithms. Kulhmann and colleagues performed rigorous statistical evaluation including correction for multiple comparisons for the crowd-sourced algorithms that were obtained via the ‘Melbourne-University AES-MathWorks-NIH Seizure Prediction Challenge’ conducted at kaggle.com. Their results showed that no one algorithm is the best, rather different algorithms perform optimally for different patients, leading to the view that patient-specific algorithms should be used [2].
Some Fundamental Challenges
There are a couple fundamental challenges that need to be overcome that might aid in making seizure prediction a reality
- Human ‘Expert’ in the loop. Predicting events in the EEG that have been classified by humans based on subjective criteria has inherent challenges. Consider that these subjective events have to be described by one expert to another in non-quantitative terms to be ‘learned’ while the quantitative descriptions of these subjective ‘eyeball’ judgements have to then be ‘discovered’ using algorithms. Consequently, what one expert marks as a seizure is not necessarily what another one marks as a seizure. There have been a few studies comparing different experts, for e.g. [4] which arrived at correspondence of 80% overlap. However typically these experts come from one school of subjective description. Comparing a larger pool across hospitals or research groups may result in much lower correlation. Further, in cases of high seizure rates and as well as long and short duration seizures, there could be considerable disagreement between the experts [3].
- Correspondence between EEG and other seizure symptoms. There is also the challenge that there is no clear understanding of how ‘expert’ coded seizures in the EEG relate to other mechanisms of seizure identification such as muscle stiffness or loss of consciousness. Seizures come with a wide range of symptoms making this correspondence challenging. The lack of substantial data drawing this correspondence thus adds further subjectivity to the marking of ‘seizures’ in the EEG.
In the ideal circumstance EEG is measured along with annotations of clinical symptoms of seizure in real time allowing a direct analysis of the structure of the EEG corresponding to onset of clinical symptoms. This is difficult to do however, and few datasets of this nature exist that allow bypassing an intermediate subjective assessment of the EEG.
References
- Kuhlmann, Levin, et al. “Seizure prediction—ready for a new era.” Nature Reviews Neurology14.10 (2018): 618-630.
- Kuhlmann, Levin, et al. “Epilepsyecosystem. org: crowd-sourcing reproducible seizure prediction with long-term human intracranial EEG.” Brain141.9 (2018): 2619-2630.
- Baumgartner, Christoph, Johannes P. Koren, and Michaela Rothmayer. “Automatic computer-based detection of epileptic seizures.” Frontiers in neurology9 (2018): 639.
- Wilson, S, et al. Seizure Detection: Correlation of Human Experts Clin Neurophysiology (2003)
Further links